
Generics are scientifically identical to brand-name drugs, yet many people believe they don't work as well. This article explains why the perception gap exists, how it affects health outcomes, and what really works to build trust.

Generics are scientifically identical to brand-name drugs, yet many people believe they don't work as well. This article explains why the perception gap exists, how it affects health outcomes, and what really works to build trust.
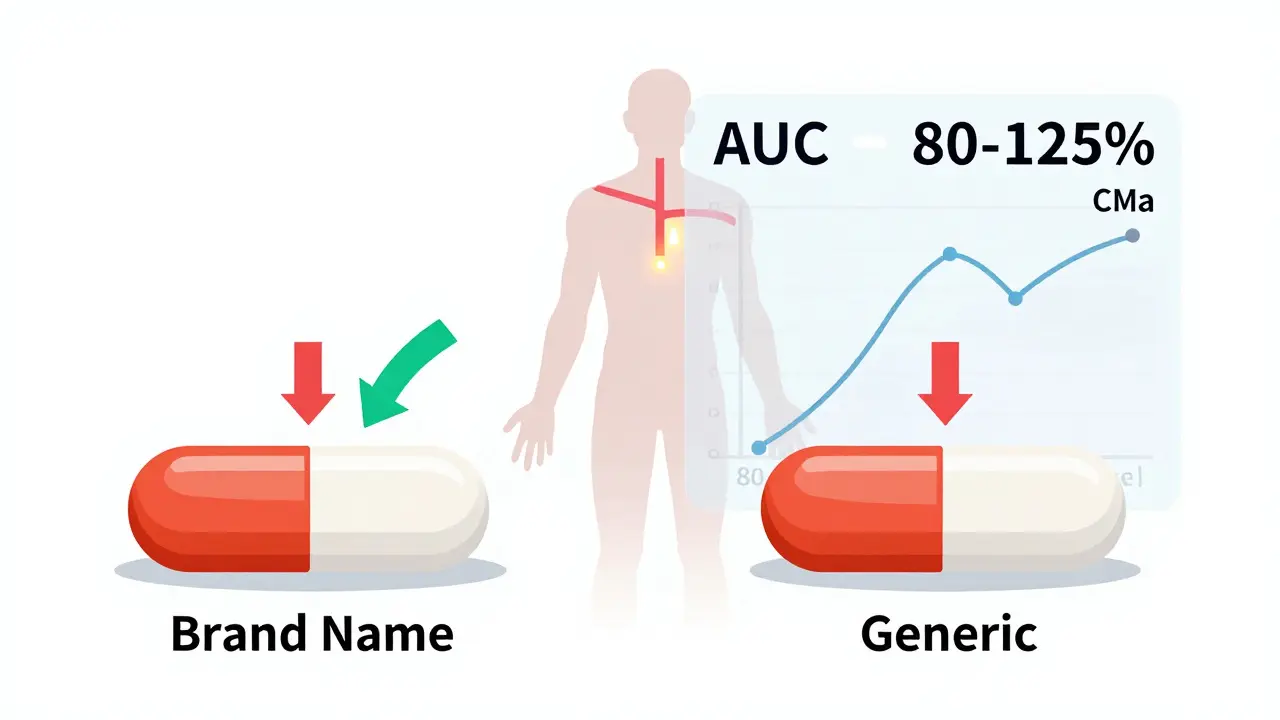
Bioequivalence studies are the scientific foundation that ensures generic drugs work the same as brand-name versions. The FDA requires strict pharmacokinetic testing to prove identical absorption and effectiveness - a process that keeps patients safe and lowers drug costs.

Doctors around the world view generic medications differently based on healthcare systems, costs, and policies. From Europe's mandatory substitution to Asia's life-saving access, generics are reshaping global care.
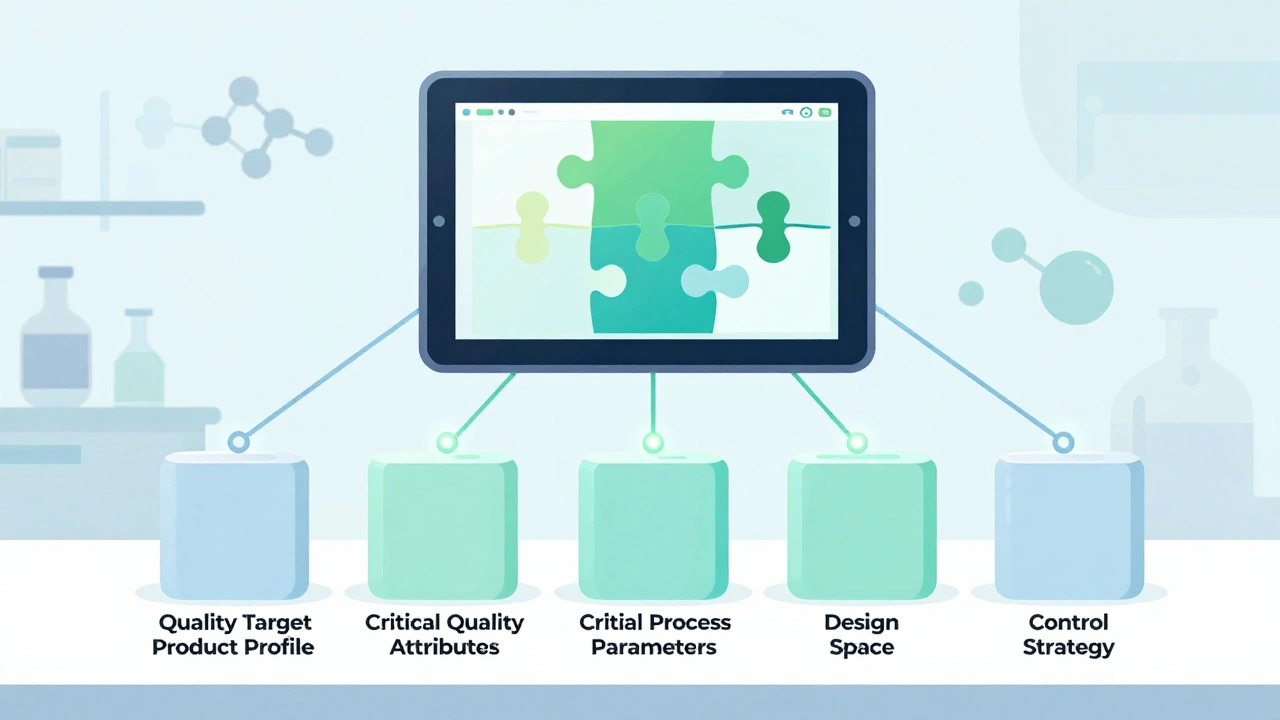
Quality by Design (QbD) has transformed generic drug development by replacing guesswork with science. Learn how QbD improves approval rates, cuts review times, and ensures bioequivalence without clinical trials - backed by FDA data and real-world case studies.

The Hatch-Waxman Amendments of 1984 created the modern generic drug system in the U.S., cutting costs by up to 90% and boosting generic market share to 90%. It balanced innovation incentives with patient access-and still shapes drug prices today.