How TRIPS Patent Rules Shape Global Access to Generic Medicines
 Mar, 17 2026
Mar, 17 2026
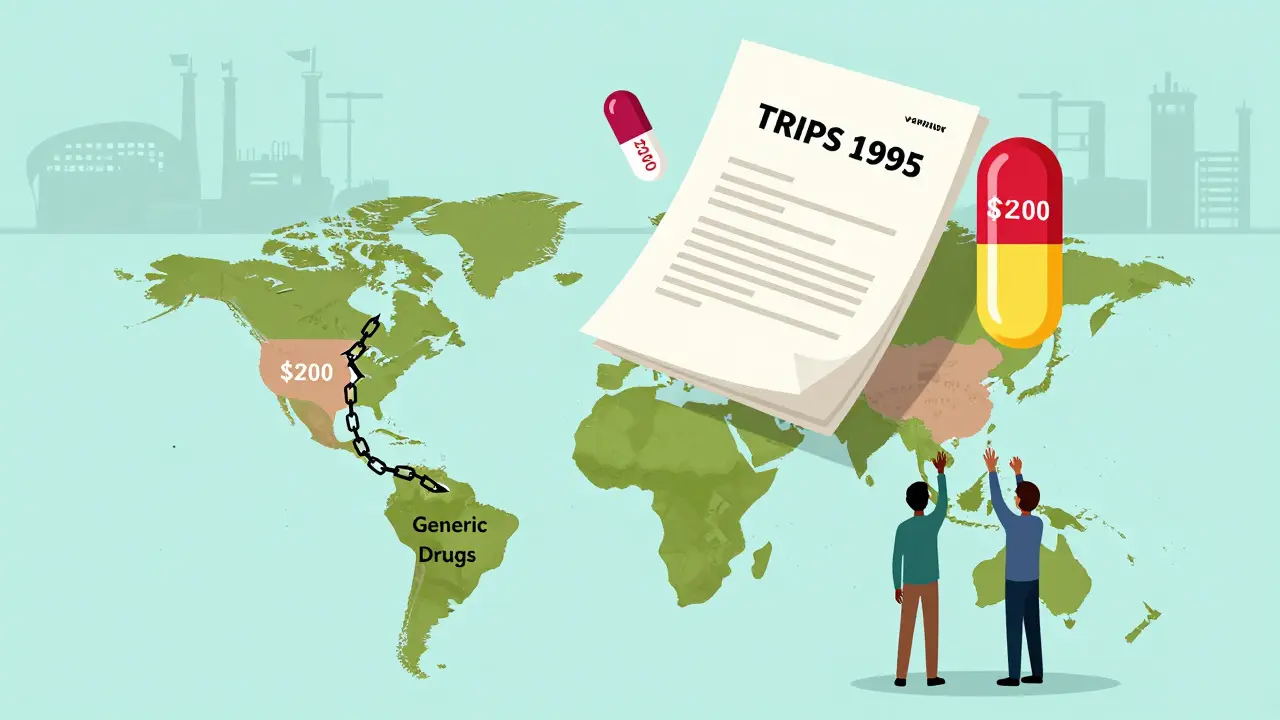
When you take a generic version of a blood pressure or HIV drug, you might think it's just cheaper medicine. But behind that pill is a decades-long battle over patent policy, international trade rules, and who gets to live - and who gets left behind. The TRIPS Agreement is a global legal framework under the World Trade Organization that sets minimum standards for patent protection, including for pharmaceuticals. It came into force in 1995 and changed everything about how generic drugs are made, sold, and distributed around the world.
What TRIPS Actually Does to Generic Drugs
Before TRIPS, many developing countries didn’t even allow patents on drugs. They could make their own versions using different manufacturing methods - called process patents - and sell them cheaply. India, for example, was a global hub for generic HIV and cancer drugs because it only protected the process, not the final product. That meant a drug costing $10,000 a year in the U.S. could be made in India for $200. But TRIPS changed that. It forced every WTO member - 164 countries - to grant product patents on medicines for at least 20 years from the filing date. No more shortcuts. No more workarounds. If a drug is patented, you can’t make it, even if you use a different method.
That 20-year rule sounds fair - until you realize it starts counting from when the patent is filed, not when the drug hits the market. Many drugs sit in regulatory review for years before approval. So by the time a generic version can legally enter the market, the effective monopoly might already be 25 years long. Add in data exclusivity - where regulators can’t use the original company’s clinical trial data to approve generics - and you’re looking at 5 to 10 more years of no competition. That’s why, in places like South Africa and Brazil, prices for new HIV drugs didn’t drop for over a decade after TRIPS kicked in.
The Compulsory License Loophole (That Almost Doesn’t Work)
TRIPS does have a built-in escape hatch: compulsory licensing. Article 31 says a government can force a patent holder to let someone else make the drug - without permission - if there’s a public health emergency. Sounds great, right? But here’s the catch: the license can only be used to supply the domestic market. So if Nigeria needs 10 million doses of a cancer drug but can’t produce it, it can’t import cheaper versions from India. India can’t export under its own compulsory license because TRIPS says it has to be for its own people.
This rule was a disaster for countries without drug factories. In 2001, the Doha Declaration tried to fix it by affirming that public health trumps patents. But it didn’t change the law - just said countries could interpret it more flexibly. The real fix came in 2005: the "Paragraph 6 Solution," which allowed countries to export generics made under compulsory license. Sounds like progress? Not really. Only two countries ever used it: Canada and Rwanda. Canada made a small shipment of HIV drugs to Rwanda in 2007. That was it. The paperwork was too complex. The legal risks too high. The pharmaceutical industry too loud. By 2016, only one shipment of malaria medicine had been sent under this rule.
TRIPS Plus: The Hidden Rules That Hurt More
Even if a country follows TRIPS exactly, it’s still getting squeezed. That’s because rich countries don’t stop at TRIPS. They push "TRIPS Plus" rules into bilateral trade deals. The U.S., EU, and Switzerland routinely demand extra protections in exchange for market access. These include:
- Extending patent terms beyond 20 years
- Imposing data exclusivity periods longer than TRIPS allows
- Banning parallel imports (buying cheaper versions from other countries)
- Requiring patent linkage - where drug regulators must check patent status before approving generics
By 2020, 85% of U.S. free trade agreements included these stricter rules. The EU-Vietnam deal, signed in 2020, gave eight years of data exclusivity - four years longer than TRIPS permits. Countries like Thailand and Brazil tried to use compulsory licenses for HIV drugs. They were hit with threats of trade sanctions. The message was clear: follow our rules, or face consequences.

Who Pays the Price?
The numbers don’t lie. In 1995, only 60 developing countries had product patents on medicines. By 2010, that number jumped to 147. The result? Prices for patented drugs soared. A 2001 study in the Journal of the American Medical Association found average price increases of over 200% in developing countries. In India, after switching to product patents in 2005, the cost of some cancer drugs jumped 300-500%. The World Health Organization found that 70% of new drugs approved between 1975 and 1997 were for diseases affecting rich countries - and only 13 were for tropical diseases like malaria or sleeping sickness. The market didn’t just ignore these diseases; it actively punished countries that tried to fix them.
Meanwhile, generic manufacturers in India, Brazil, and South Africa - once the backbone of global access - now face legal threats, patent lawsuits, and export bans. In 2001, 40 pharmaceutical companies sued South Africa over its law allowing generic imports. The case was dropped only after global protests. Brazil produced generic antiretrovirals in 2000 and got threatened with U.S. trade sanctions under Section 301. It worked - the U.S. backed down, but the message was sent: don’t challenge patents.
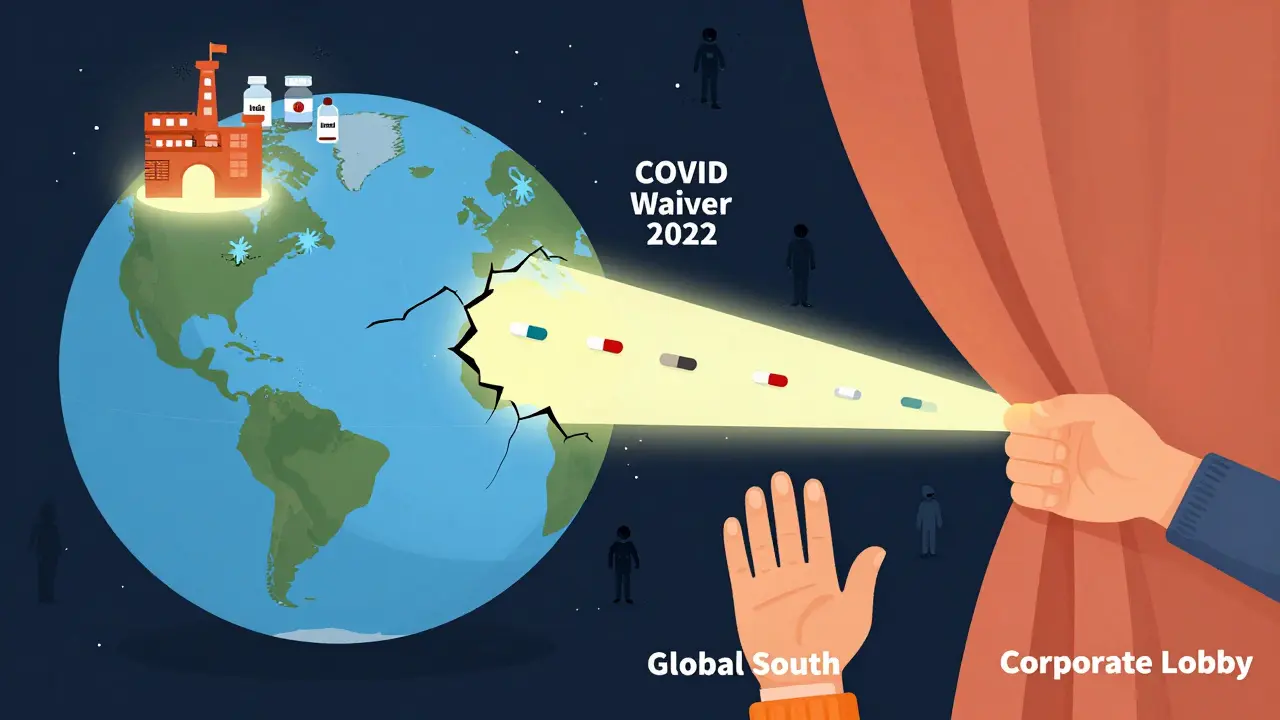
What Changed After COVID-19?
The pandemic forced the world to look at TRIPS again. In October 2020, India and South Africa proposed a temporary waiver of TRIPS protections for COVID-19 vaccines, tests, and treatments. Over 100 countries supported it. The U.S., EU, and Switzerland opposed it. The fight lasted over a year. In June 2022, the WTO agreed to a partial waiver - but only for vaccines, not treatments or diagnostics. And even that waiver has strings: countries must notify the WTO, and the waiver expires in 2025. It’s a symbolic win, but not a structural fix. The same system that blocked access to HIV drugs in the 2000s is still in place today.
The Real Winners and Losers
On one side, pharmaceutical companies argue that strong patents drive innovation. They point out that 73% of new medicines since 2000 came from companies in countries with strong IP protection. That’s true - but it’s also true that 80% of those drugs are for conditions affecting wealthy populations. The same companies that lobby for patent extensions in Africa are not investing in drugs for neglected diseases. The Medicines Patent Pool, a nonprofit that negotiates voluntary licenses with drugmakers, has helped 17.4 million people get HIV, hepatitis C, and TB treatments since 2010. But it only works because companies choose to license - not because they’re forced to.
On the other side, countries with weak manufacturing capacity - like most of sub-Saharan Africa - are stuck. They can’t make generics. They can’t import them legally. And they can’t afford the branded versions. The Global Fund reports that while first-line HIV treatment dropped from $10,000 per patient per year in 2000 to $75 in 2019, second-line drugs still cost $300-$600. That’s because second-line drugs are newer, patented, and protected by data exclusivity. No generics yet. No competition. No price drop.
Is There a Way Forward?
There are solutions - but they require political will, not just legal tweaks. Countries need to:
- Use TRIPS flexibilities aggressively - compulsory licensing, parallel imports, and exceptions for public health
- Build regional manufacturing hubs - like the African Medicines Agency - to reduce dependence on imports
- Reject TRIPS Plus in trade deals - no more data exclusivity, no more patent term extensions
- Push for a full TRIPS waiver on essential medicines - not just during pandemics, but permanently
India and South Africa proved it’s possible: they produced life-saving drugs at scale. But they were punished for it. The system isn’t broken - it was designed this way. To protect profits. To delay competition. To keep prices high. Until that changes, millions will keep dying not because medicine doesn’t exist - but because the rules say they can’t have it.
Does TRIPS ban all generic drugs?
No, TRIPS doesn’t ban generic drugs. It bans the production of generics before a patent expires. But it allows countries to issue compulsory licenses for public health emergencies, and to import generics from other countries under limited conditions. The problem isn’t the law - it’s how it’s enforced and the extra rules (TRIPS Plus) that many countries are forced to accept.
Why can’t poor countries just make their own generics?
Many can - but they’re blocked by patent laws, lack of manufacturing infrastructure, and pressure from rich countries. Countries like India and Brazil have strong generic industries, but they’re often targeted with trade threats if they export. Most low-income countries don’t have the factories, regulatory systems, or technical skills to produce complex medicines like biologics. Even if they could, TRIPS rules and TRIPS Plus provisions make it legally risky.
What’s the difference between a patent and data exclusivity?
A patent gives the original company exclusive rights to make and sell the drug for 20 years. Data exclusivity is separate: it prevents drug regulators from using the original company’s clinical trial data to approve a generic version - even if the patent has expired. This can add 5-10 more years of monopoly. So a drug might be off-patent, but generics still can’t enter the market because regulators can’t review the safety data.
Did the COVID-19 TRIPS waiver help?
It helped a little - but not enough. The 2022 waiver only applied to vaccines, not treatments or diagnostics. It’s temporary, expires in 2025, and requires complex notifications. No country has used it yet. The real issue remains: the system still prioritizes corporate control over public need. A true solution would be a permanent, broad waiver for all essential medicines.
Are generic drugs safe?
Yes. Generic drugs must meet the same quality, strength, and safety standards as brand-name drugs. The WHO and FDA both certify generics. In fact, 80% of all medicines taken in the U.S. are generics. The only difference is price - not quality. The fear that generics are inferior is mostly marketing - not science.

Amadi Kenneth
March 17, 2026 AT 13:53Let me tell you something no one else will: the WHO, WTO, and Big Pharma are all one giant shadow corporation-same board, same investors, same private jets. TRIPS? It was never about innovation-it was about control. They invented "data exclusivity" to keep generics out, even after patents expire. And don't get me started on how the U.S. threatens countries with trade wars just for trying to save lives-this isn't capitalism, it's pharmaceutical feudalism. I've seen the emails-yes, I have sources-there's a backdoor deal between Pfizer and the EU that bans any African nation from even *thinking* about importing generics. They call it "IP protection." I call it mass murder with a spreadsheet.
Alexander Pitt
March 17, 2026 AT 15:04The core issue is structural, not just legal. TRIPS flexibilities exist on paper, but enforcement is impossible without political power. Countries without manufacturing capacity or diplomatic leverage can't use compulsory licenses effectively. The real barrier isn't the law-it's the asymmetry of power. India and Brazil succeeded because they had industrial capacity and domestic political will. Most nations don't. The solution isn't more loopholes-it's dismantling the entire system that ties medicine access to GDP.
Manish Singh
March 18, 2026 AT 00:40I grew up in Mumbai watching my uncle’s factory make generic HIV drugs for $150 a year. Then TRIPS hit. Suddenly, our machines were illegal. We didn’t stop-we just went underground. My uncle got arrested twice. We still made pills. People died waiting for patents to expire. Now? The same drugs cost 10x because the big pharma lobbyists got their way. But here’s the truth: we still make 70% of the world’s generics. We’re not victims-we’re survivors. And if you think the system is fair, you’ve never tried to buy a cancer drug in rural India.
Nilesh Khedekar
March 19, 2026 AT 12:55you know what really happened? the usa didn't just push TRIPS... they brainwashed the whole world into thinking patents = progress. but if you look at the real stats, 80% of new drugs are for baldness, erectile dysfunction, or weight loss. not malaria. not TB. not cholera. and yet, we're told we need 20-year monopolies? lol. also, did you know that the first HIV drug patent was filed in 1987? but it didn't hit the market until 1996? so by the time generics could even start, the clock was already at 9 years. that's not innovation-that's theft with a law degree.
Robin Hall
March 19, 2026 AT 14:35It is my considered opinion, based on extensive review of WTO documentation, that the invocation of Article 31 of the TRIPS Agreement constitutes an extraordinary and legally precarious instrument of state intervention, the misuse of which may trigger international liability under customary international law. The 2005 Paragraph 6 Solution, while technically compliant, imposes an administrative burden so onerous as to render it functionally inert. Moreover, the conflation of public health imperatives with economic sovereignty undermines the foundational legitimacy of the multilateral trading system.
jared baker
March 19, 2026 AT 16:10Think of it like this: if you invent a toaster, you get to be the only one selling it for 20 years. But if it's a life-saving drug, you're not just selling a toaster-you're selling oxygen. The system says you can't let anyone else make it, even if you're the only one who can afford it. That's not capitalism. That's cruelty with a patent number.
Andrew Mamone
March 21, 2026 AT 00:30There’s a quiet revolution happening in places like Kenya and Bangladesh-local labs are starting to make biosimilars without corporate backing. It’s slow, messy, and underfunded-but it’s real. The system is rigged, yes. But people are building alternatives anyway. We don’t need permission to save lives. We just need the will. And maybe, just maybe, that’s the real loophole.
MALYN RICABLANCA
March 22, 2026 AT 01:47OMG. I just read this and I’m literally crying. Like, imagine being a mother in Lagos and your kid needs a $10,000 drug but you make $2 a day. And then some lawyer in Geneva says "sorry, patent pending." That’s not policy-that’s a horror movie. And don’t even get me started on how the EU is still pushing TRIPS Plus in Africa like it’s 1998. I’m so angry right now I could scream. Also, I just signed a petition. You should too. #TRIPSisTerrorism
gemeika hernandez
March 23, 2026 AT 08:11Everyone’s overcomplicating this. It’s simple: if a drug saves lives, it shouldn’t be patented. Period. The fact that we even debate this shows how messed up our system is. Big Pharma doesn’t care about sick people. They care about stock prices. And governments? They’re just their lobbyists in suits. Wake up. This isn’t about law. It’s about greed.
Srividhya Srinivasan
March 24, 2026 AT 11:53Let me be clear: India didn’t "break" the rules-they exploited a gray area. And now that the rules are locked in, they’re being punished for it. But here’s the real scandal: the U.S. and EU never had to make their own generics. They outsourced production to places like India, then turned around and banned them. That’s not trade-it’s colonialism with a trademark.
Justin Archuletta
March 24, 2026 AT 16:01People are dying because of paperwork. That’s insane. We need to cut the red tape. Now.
Sanjana Rajan
March 25, 2026 AT 22:11Look, I get it. Patents are supposed to incentivize innovation. But when the incentive is $200 billion in profits from one drug while millions die, something’s broken. And no, "TRIPS Plus" isn’t just a buzzword-it’s a weapon. Countries are being blackmailed into giving up their right to health for the sake of "market access." It’s not economics. It’s extortion.
Kyle Young
March 27, 2026 AT 13:27If we accept that life is a commodity, then the patent system is merely a rational mechanism for allocating scarce resources. But if we reject that premise-if we believe health is a human right-then the entire framework collapses. The question isn’t whether TRIPS is legally sound. It’s whether we are morally willing to live in a world where a child’s survival depends on a corporate balance sheet.
Aileen Nasywa Shabira
March 28, 2026 AT 07:33Oh wow, a 10-page essay about how rich people are mean. Groundbreaking. Next you’ll tell me oxygen is expensive in Antarctica. Maybe if these countries stopped being so lazy and built their own labs instead of whining about "TRIPS Plus," they wouldn’t be in this mess. Also, I heard the WHO is funded by Gates and Pfizer. Coincidence? I think not.
lawanna major
March 28, 2026 AT 19:11There’s a deeper truth here: the pharmaceutical industry doesn’t just profit from illness-it depends on it. If generics were universally accessible, their entire business model collapses. That’s why they fund think tanks, lobby legislators, and pressure the WHO. This isn’t about innovation-it’s about sustaining a system that thrives on dependency. The real solution isn’t legal reform-it’s economic reimagining. We need public drug development, not corporate monopolies. And yes, it’s possible. We just need the courage to demand it.